Pharmacy manufacturer rebate negotiation strategies: A common ground for a common purpose
Pharmaceutical manufacturers and Medicare Part D prescription drug plan (PDP) sponsors have a common interest in serving the health needs of Medicare beneficiaries. However, companies from both industries are subject to competitive pressures and financial performance expectations and often find themselves at opposite ends of the table in tense negotiations. This article explores the objectives of PDP sponsors and pharmaceutical manufacturers and proposes a common ground where both can prosper and work together to meet the needs of the beneficiaries who rely on them.
PDP sponsors operate in a highly competitive market and must effectively control the cost of providing benefits to attract and retain members. To remain competitive, PDP sponsors aggressively manage the lists of drugs covered by their plans on their formularies. There are often numerous ways to structure formulary coverage that will provide appropriate drug therapy choices for prescribing physicians. PDP sponsors must consider the net financial cost of a product relative to other available products when making formulary placement decisions. The net financial cost reflects all pharmacy discounts, beneficiary cost sharing, and rebates paid by pharmaceutical manufacturers.
Pharmaceutical manufacturers prosper through the sales of their products. Members are more likely to take products if they are covered by their plans’ formularies. To increase sales, pharmaceutical manufacturers pay rebates to PDP sponsors in exchange placing their products on formularies, improving their tier placements, and avoiding dispensing barriers like step edits or prior authorization requirements.
Rebate negotiations are critically important for both PDP sponsors and pharmaceutical manufacturers. Insufficient rebates may make PDP sponsors uncompetitive while excessive rebates will deteriorate pharmaceutical manufacturers’ profits.
PDP sponsors calculate the rebate level necessary to make the inclusion of a particular drug (or adoption of a particular set of drugs for a therapeutic class) financially advantageous. If the pharmaceutical manufacturer is willing to pay that rebate level, then an agreement can often be reached and both parties will benefit. The graphic in Figure 1 shows the common ground and the range of potential outcomes from negotiations.
Figure 1: Common Ground Illustrated
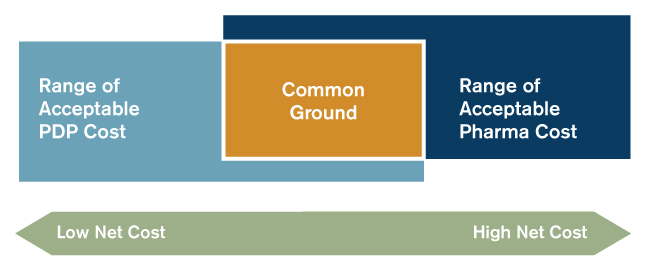
Both PDP sponsors and pharmaceutical manufacturers should enter negotiations with well-developed estimates of the range depicted in Figure 1. It is important that both parties understand the range from their own perspectives as well as from the perspective of the other party. A variety of factors influences the PDP acceptable cost range, including the mix of low income and non-low income members, the structure of their formularies, and formulary coverage alternatives as well as their product portfolios and marketing strategies. Pharmaceutical manufacturers' acceptable cost ranges are influenced by their pricing strategies, their abilities to find alternate distribution channels, and their tolerance for lost volume. When one side does not have a good understanding of the other side’s acceptable range, it is likely that either one party could overpay or that the parties will not arrive at agreeable terms. Rather than let such an outcome occur, both parties should consider seeking assistance in determining the cost range.
Figure 2: Example of Net Cost With 2016 Price Protection
| 1/1/2016 | 3/1/2016 | 12/1/2016 | 1/1/2017 | |
| Drug cost | $100.00 | $105.00 | $115.00 | $115.00 |
| Rebates | $30.00 | $31.50 | $31.50 | $34.50 |
| Price Protection | $0.00 | $0.00 | $10.00 | $0.00 |
| Net Cost | $70.00 | $73.50 | $73.50 | $80.50 |
| Net PDP Liability1 | $39.00 | $40.95 | $39.95 | $44.85 |
Once common ground is found and a deal is reached, the agreement could be relatively short-lived if the cost of the drug increases significantly as we have seen in recent years. The 2015 Milliman Medical Index estimates that the increase in pharmacy claims costs due to unit cost increases was 13% between 2014 and 2015. This high trend jeopardizes a drug’s position on a formulary because it might result in other drugs becoming more cost-effective while still providing appropriate prescribing options for physicians.
In recent years, price protection provisions have become more common in rebate contracts and have evolved into an important part of providing financial protection for PDP sponsors. Price protection arrangements provide a cost threshold above which additional rebate payments will be made. For example, a drug with a $100 cost might have a 30% rebate with price protection that occurs after a 5% increase in cost. If the drug price increases to $115, the rebate would total 30% of $105 plus $10 (see Figure 2).
In this example, the manufacturer is responsible for 100% of the overage above the price threshold. Price protection can expire at the end of a year leaving the price of $115 as the starting point with only a 30% rebate. This represents the full 15% increase in net PDP liability when price protection lapses and would often cause a PDP sponsor to reconsider the formulary placement of the drug and evaluate alternative coverage options within the therapeutic class. Some price protection agreements run over multiple years or renegotiate subsequent years at higher rebates, however.
Stability is needed to form a long-term relationship between the PDP and pharmaceutical manufacturer to increase the likelihood of longer-term and predictable formulary placement. The foundation of this long-term relationship could include:
- The two parties agree to a baseline rebate amount (e.g., 30%) and a price protection threshold. PDP sponsors should seek low thresholds (e.g., 2%) in order to help control the cost of providing benefits throughout the plan year.
- Price protection may be set for a percentage of the overage. For example, 90% might be enough to protect the PDP sponsor against net liability increases because the full rebate increase is not always needed since the PDP net cost of the drug varies by the coverage band (e.g., in the coverage gap a PDP pays 5% of the drug cost in 2016 but receives all of the rebate). This provides a revenue increase for the pharmaceutical manufacturer while still providing a financial hedge against increasing pharmacy costs for the PDP sponsor.
- At year-end, the rebate is not reset to the original level but rather is set such that the pharmaceutical manufacturer yields a predetermined net revenue increase (e.g., 4%). This allows the PDP sponsor to retain the drug at its existing level without the need to reevaluate the therapeutic class and consider changing coverage because of double-digit cost increases. See Figure 3 for an example.
- Pharmaceutical manufacturers are sometimes blindsided with formulary changes and do not find out about them until after the decisions to exclude their drugs have been made. Part of the agreement may include a clause whereby the PDP sponsor must notify the manufacturer at least 30 days in advance of dropping a drug so that there is an opportunity for a constructive dialogue, which might influence the final coverage decision.
This structure is not intended to create binding legal restrictions but rather is intended to codify an arrangement that is mutually beneficial by helping each side to achieve its objectives. Under this proposed structure, the PDP sponsor achieves cost control with less earnings volatility while the manufacturer achieves increased volume and regular revenue increases. In addition, the resulting stability better serves the needs of Medicare beneficiaries by creating continuity and could improve the quality of care and service.
Figure 3: Baseline Rebate Increases in 2017 to Yield 4% Increase
| 1/1/2016 | 3/1/2016 | 12/1/2016 | 1/1/2017 | |
| Drug cost | $100.00 | $102.00 | $115.00 | $115.00 |
| Rebates | $30.00 | $30.60 | $30.60 | $42.20 |
| Price Protection | $0.00 | $0.00 | $11.70 | $0.00 |
| Net Cost | $70.00 | $71.40 | $72.70 | $72.80 |
| Net PDP Liability1 | $39.00 | $39.78 | $39.39 | $39.46 |
Important disclosures
This communication has been prepared for the specific purpose of demonstrating how PDPs and pharmaceutical manufacturers can reach a common ground in rebate negotiations. This information may not be appropriate, and should not be used, for any other purpose.
Milliman does not intend to benefit or create a legal duty to any third-party recipient of its work. This communication must be read in its entirety.
Milliman does not provide legal advice and recommends that others consult with their legal advisors regarding legal matters.
Contact
If you have any questions or comments on this document, please contact Steve Kaczmarek, who is a principal and consulting actuary with Milliman. Steve is a member of the American Academy of Actuaries and meets the Qualification Standards of the American Academy of Actuaries to render the actuarial opinion contained herein.
Steve Kaczmarek
[email protected]
+1 860 687 0121
About the Author(s)
Stephen Kaczmarek
Pharmacy manufacturer rebate negotiation strategies: A common ground for a common purpose
This article explores the objectives of Medicare Part D prescription drug plan sponsors and pharmaceutical manufacturers and proposes a common ground where both can prosper and work together to meet the needs of beneficiaries.